The FDA Says Your Pharmacy Can Legally Give You Abortion Pills Now—If It’s Willing
There are some small administrative hoops to jump through, but mifepristone could soon come to a drug store near you.
AbortionPolitics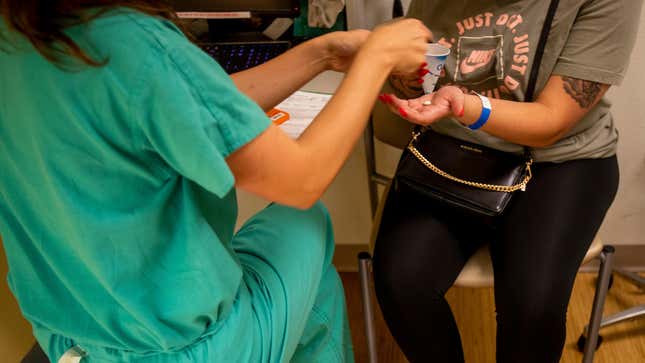
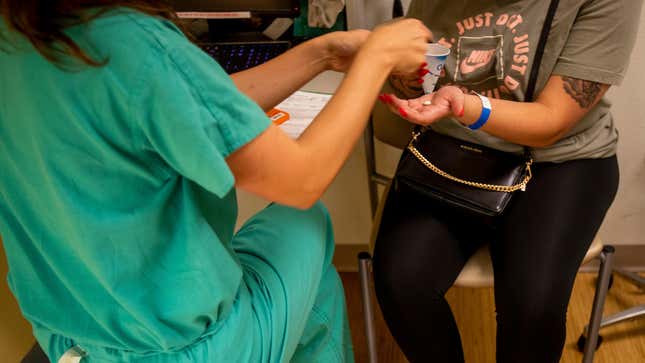
Retail pharmacies—both local and national chains—may legally dispense mifepristone, the first of two drugs in medication abortions, according to a new ruling from the FDA released on Tuesday. The guidance is the latest move by the regulatory agency to increase access to medication abortion.
However, this doesn’t mean you’ll be able to find a box of mifepristone in the aisles of CVS anytime soon. Under the new ruling, patients still need a certified provider to write a prescription for mifepristone, and the pharmacy must meet some administrative requirements. It’s unclear how many retail chains will go through this extra step without being pushed by consumers. (The second drug commonly used in medication abortion regimens, misoprostol, is used for other medical purposes and already accessible via prescription.)
-

-

-

-

-

-

-

-

-

-

-

-

-

-

-

-

-

-

-

-

-

-

-

-

-

-

-

-

-

-

-

-

-

-

-

-

-

-

-

-